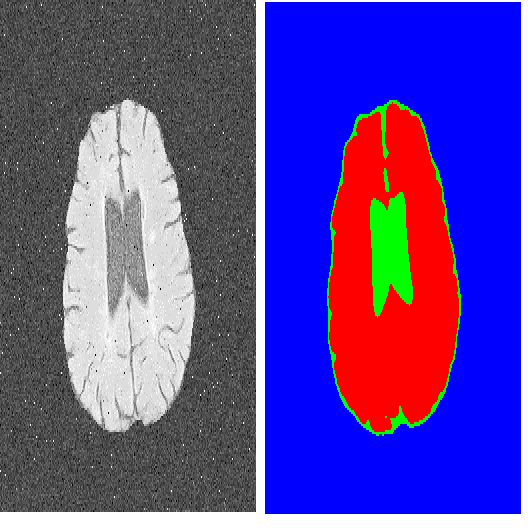
Image segmentation is the process of partitioning the image into significant regions easier to analyze. Nowadays, segmentation has become a necessity in many practical medical imaging methods as locating tumors and diseases. Hidden Markov Random Field model is one of several techniques used in image segmentation. It provides an elegant way to model the segmentation process. This modeling leads to the minimization of an objective function. Conjugate Gradient algorithm (CG) is one of the best known optimization techniques. This paper proposes the use of the Conjugate Gradient algorithm (CG) for image segmentation, based on the Hidden Markov Random Field. Since derivatives are not available for this expression, finite differences are used in the CG algorithm to approximate the first derivative. The approach is evaluated using a number of publicly available images, where ground truth is known. The Dice Coefficient is used as an objective criterion to measure the quality of segmentation. The results show that the proposed CG approach compares favorably with other variants of Hidden Markov Random Field segmentation algorithms.
翻译:图像分割是将图像分割成更便于分析的重要区域的过程。 如今, 分离已成为许多实用医学成像方法中的一种必要条件, 例如定位肿瘤和疾病。 隐藏的 Markov 随机场模型是图像分割法中使用的几种技术之一 。 它为模拟分割过程提供了一个优雅的方法 。 这种建模可以使客观功能最小化 。 Conjugate Gradient 算法( CG) 是最已知的优化技术之一 。 本文建议使用基于隐藏的 Markov 随机场的 Conjugate Gradient 算法( CG) 进行图像分割 。 由于此表达式没有衍生物, 在 CG 算法中使用有限差异来估计第一个衍生物 。 在已知地面真相的情况下, 使用一些公开的图像来评估该方法 。 Dice Covalent 用作衡量分割质量的客观标准 。 结果表明, 拟议的 CG 方法比隐藏 Markov 随机场分割算法的其他变量要好。