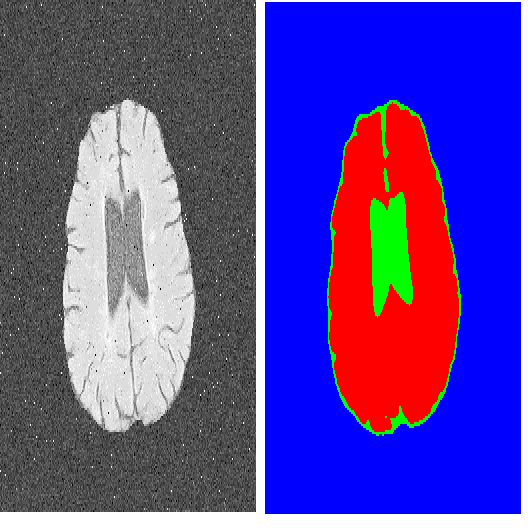
We propose an Active Learning approach to image segmentation that exploits geometric priors to streamline the annotation process. We demonstrate this for both background-foreground and multi-class segmentation tasks in 2D images and 3D image volumes. Our approach combines geometric smoothness priors in the image space with more traditional uncertainty measures to estimate which pixels or voxels are most in need of annotation. For multi-class settings, we additionally introduce two novel criteria for uncertainty. In the 3D case, we use the resulting uncertainty measure to show the annotator voxels lying on the same planar patch, which makes batch annotation much easier than if they were randomly distributed in the volume. The planar patch is found using a branch-and-bound algorithm that finds a patch with the most informative instances. We evaluate our approach on Electron Microscopy and Magnetic Resonance image volumes, as well as on regular images of horses and faces. We demonstrate a substantial performance increase over state-of-the-art approaches.
翻译:我们提议了一种积极的学习方法来利用几何前缀来简化批注过程。 我们用2D 图像和 3D 图像积分的背景前景和多级分割任务来展示这一点。 我们的方法结合了图像空间的几何平滑前期, 以及更传统的不确定措施来估计哪些像素或氧化物最需要批注。 对于多级设置, 我们还引入了两种新的不确定性标准。 在 3D 案中, 我们使用由此产生的不确定性措施来显示位于同一平板补板上的告示器, 这使得批量批量注比在卷内随机分布容易得多。 我们发现平面补板使用了分支和宽度的算法, 找到与信息最多的例子的补丁。 我们评估了我们在电子显微镜和磁振荡图像量上的方法, 以及马和面的常规图像上的方法。 我们展示了相对于最新方法的显著性能提高。